Your basket is currently empty!

FDA eSTAR: Navigating the New Era of Medical Device Submissions
Introduction To The FDA eSTAR Program
The medical device industry is witnessing a significant transformation in regulatory affairs, particularly in how manufacturers submit their applications to the US Food and Drug Administration (FDA). The Electronic Submission Template And Resource (eSTAR) program represents the FDA’s commitment to modernizing the review process for medical devices, moving away from traditional paper-based submissions toward a more streamlined, consistent digital approach.
For new manufacturers seeking to enter the US market, understanding the eSTAR program is no longer optional—it’s essential knowledge in the field of medical device regulatory compliance. As regulatory requirements continue to evolve, staying informed about these changes can make the difference between a successful market entry and costly delays.
What Is The FDA eSTAR Program?
The eSTAR program is an interactive PDF-based template designed to guide manufacturers through the preparation of 510(k) premarket submissions, De Novo requests, and Premarket Approval (PMA) applications. Launched as part of the FDA’s ongoing efforts to enhance review efficiency, eSTAR aims to:
- Streamline the submission process
- Improve consistency in applications
- Reduce review times
- Decrease the frequency of additional information requests
- Facilitate a more transparent regulatory pathway
For professionals in medical device regulatory affairs, the eSTAR program represents a significant shift in how regulatory documentation is prepared and submitted, requiring updated skills and knowledge. eSTAR has been mandatory for 510(k) submissions since October 2023 and will become mandatory for De Novo submissions from October 2025.
Advantages Of The eSTAR Program For Medical Device Manufacturers
1. Enhanced Submission Quality and Completeness
The eSTAR template provides a guided approach that helps ensure all necessary information is included before submission. This structured format significantly reduces the likelihood of incomplete applications—one of the primary reasons for submission delays.
For manufacturers with limited experience in US FDA regulations for medical devices, this guided approach serves as a valuable resource, essentially providing a roadmap through the complex submission process.
2. Accelerated Review Timelines
By standardizing the format and organization of submissions, eSTAR allows FDA reviewers to locate and assess critical information more efficiently. This standardization can potentially lead to faster review cycles and quicker time-to-market for new devices.
According to FDA data, submissions using eSTAR have demonstrated shorter overall review times compared to traditional submissions, highlighting a tangible benefit for manufacturers invested in rapid market access.
3. Reduced Additional Information Requests
The guided structure of eSTAR helps manufacturers address potential questions before submission, reducing the need for additional information requests (AIRs) during the review process. For companies engaged in medical device compliance, this means fewer regulatory hurdles and more predictable timelines.
4. Integration With FDA Digital Transformation Initiatives
The eSTAR program aligns with the FDA’s broader digital transformation strategy, positioning manufacturers who adopt it early to better adapt to future regulatory technology advancements. This forward-thinking approach is increasingly valuable in regulatory affairs masters programs and professional development.
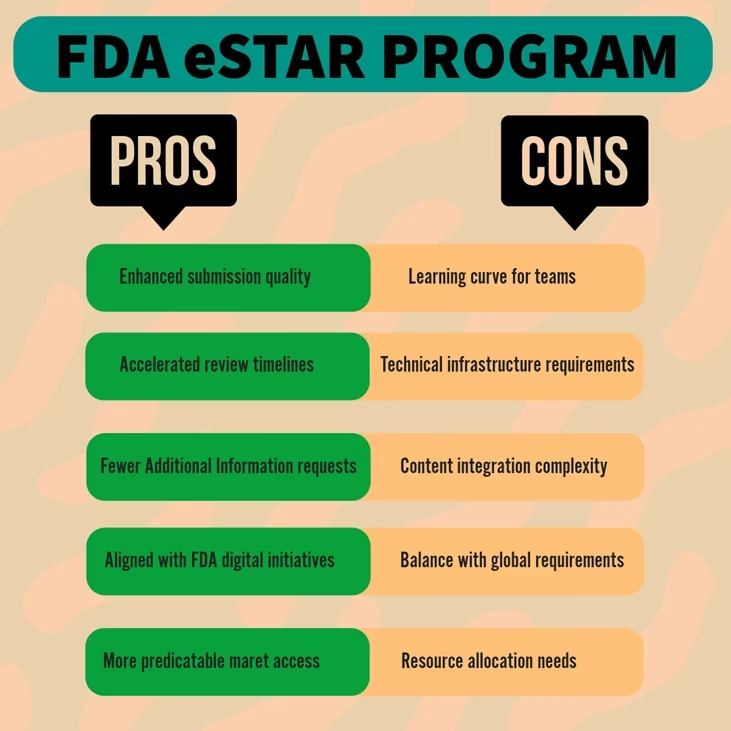
Potential Challenges and Pitfalls
Despite its advantages, the transition to eSTAR presents several challenges that manufacturers should be prepared to address:
1. Learning Curve and Resource Requirements
Adapting to the eSTAR format requires an investment in training and possibly new personnel with specific expertise. Manufacturers must ensure their regulatory teams are properly trained in medical device regulatory compliance specific to eSTAR requirements.
For smaller manufacturers or startups with limited regulatory resources, this learning curve can be particularly steep, potentially necessitating specialized medical device training courses or consultation.
2. Technical Infrastructure Needs
The eSTAR system requires certain technical capabilities, including compatible software and sufficient computing resources to handle the interactive PDF format. Organizations must assess their technical readiness before committing to eSTAR submissions.
3. Content Integration Complexities
While eSTAR standardizes the submission format, manufacturers still face the challenge of effectively integrating their existing documentation into this new structure. This may require significant reformatting and reorganization of technical documentation.
4. Balancing Global Regulatory Strategies
Manufacturers operating in multiple markets must balance eSTAR requirements with other regional submission formats, such as the European Union Medical Device Regulation (EU MDR). Developing a cohesive global strategy while maintaining compliance with US-specific requirements demands sophisticated regulatory affairs expertise.
Best Practices for Successful eSTAR Implementation
To maximize the benefits of eSTAR while minimizing potential pitfalls, manufacturers should consider the following best practices:
1. Invest in Comprehensive Training
Ensure your regulatory team receives proper training specifically focused on eSTAR requirements and processes. Consider enrolling team members in specialized regulatory affairs online courses or medical device training programs that address eSTAR specifically.
2. Develop a Transition Strategy
Create a detailed plan for transitioning from traditional submission processes to eSTAR, including timelines, resource allocation, and contingency plans. This strategic approach is often covered in advanced regulatory compliance training programs.
3. Leverage Available FDA Resources
The FDA offers various resources to support eSTAR adoption, including webinars, guidance documents, and direct assistance. Manufacturers should take full advantage of these resources before attempting their first eSTAR submission.
4. Implement Quality Control Measures
Develop specific quality control processes tailored to eSTAR submissions to ensure accuracy and completeness before final submission. This aspect of quality management is a critical component of medical device regulatory affairs expertise.
5. Consider Regulatory Affairs Partnerships
For organizations with limited internal regulatory capability, partnering with experienced regulatory consultants or firms specializing in US FDA training courses and submissions can significantly smooth the transition to eSTAR.
The Future of Medical Device Submissions
The eSTAR program represents just one aspect of the FDA’s ongoing efforts to modernize the regulatory process for medical devices. As digital transformation continues to reshape regulatory affairs, manufacturers can expect:
- Further integration of artificial intelligence in submission review processes
- Enhanced data exchange capabilities between manufacturers and regulatory authorities
- Evolution toward a fully digital submission ecosystem
- Greater harmonization between US requirements and global medical device regulations
For professionals in the field, staying current through continuous education, such as regulatory affairs online programs and specialized medical device regulation courses, will be essential to navigating this evolving landscape.
Conclusion: Preparation is Key
The FDA’s eSTAR program offers significant advantages for medical device manufacturers seeking US market access, from improved submission quality to potentially faster review times. However, realizing these benefits requires proper preparation, training, and strategic planning.
For regulatory professionals and manufacturers alike, investing in comprehensive regulatory affairs courses and medical device compliance training is no longer optional—it’s a strategic necessity in an increasingly complex regulatory environment.
Whether you’re a seasoned regulatory professional or new to the field, deepening your understanding of eSTAR and broader US FDA regulations for medical devices will position you for success in this new era of digital submissions.
Leave a Reply